

Copyright © 2010
[Posted October 31, 2006]
Update July 1, 2010
Abstract
Even the simplest currently living cells contain hundreds of proteins most of which are essential to their functioning. Yet such complexity cannot have stood at the origin of life. Based on research in the field it is proposed here how, once a self-replicating genetic molecule existed, life might have started and gradual evolution of complexity was made possible in contrast to the sudden appearance of complexity that creationists claim to have been necessary at the beginning of life. Conditions for synthesis of organic molecules on the early Earth are reviewed, and gene-first and metabolism-first models are discussed. While the origin of the homochirality of amino acids and sugars has been a puzzling problem for decades, recent findings provide plausible explanations.
Other Links:
|
Contents
![]() cience
shows us that the universe evolved by self-organization of matter towards more
and more complex structures. Atoms, stars and galaxies self-assembled out of
the fundamental particles produced by the Big Bang. In first-generation stars, heavier
elements like carbon, nitrogen and oxygen were formed. Aging first-generation
stars then expelled them out into space we, who consist of these elements,
are thus literally born from stardust. The heaviest elements were born in the
explosions of supernovae. The forces of gravity subsequently allowed for the
formation of newer stars and of planets. Finally, in the process of biological
evolution from bacteria-like tiny cells (the last universal common ancestor,
abbr. LUCA) to all life on earth, including us humans, complex life forms arose
from simpler ones.
cience
shows us that the universe evolved by self-organization of matter towards more
and more complex structures. Atoms, stars and galaxies self-assembled out of
the fundamental particles produced by the Big Bang. In first-generation stars, heavier
elements like carbon, nitrogen and oxygen were formed. Aging first-generation
stars then expelled them out into space we, who consist of these elements,
are thus literally born from stardust. The heaviest elements were born in the
explosions of supernovae. The forces of gravity subsequently allowed for the
formation of newer stars and of planets. Finally, in the process of biological
evolution from bacteria-like tiny cells (the last universal common ancestor,
abbr. LUCA) to all life on earth, including us humans, complex life forms arose
from simpler ones.
Upon considering this self-organization of material structures in the realm of philosophy, one may conclude that it happens either because the underlying laws of nature, which have to be exceedingly special to allow for it (Rees 2001, Smolin 1999, Susskind 2006), simply are the way they are (possibly in the context of a multiverse) or because they were designed by God for this purpose. Since we know that the laws of nature are so self-sufficient that, based on them, the complexity of the entire physical universe evolved from fundamental particles, and further, complex life forms from simpler ones during biological evolution, we can reasonably extrapolate that they would also allow life itself to originate spontaneously, by chemical evolution of suitable structures regardless if we believe these laws are designed or undesigned. Therefore, we should expect an origin of life by natural causes from both theistic and atheistic philosophical perspectives.
The experimental study of the origin of life kick-started with Millers prebiotic soup experiment (Miller 1953 , Miller-Urey experiment) which produced amino acids, essential to life. In the following decades, much impressive chemistry on the building blocks of life has been performed, but for a long time many crucial questions had been without experimental answers that might give hope for firm future directions. Among these were the synthesis of nucleotides, polymerization of nucleotides to oligonucleotides, incorporation of a self-copying gene into simple cells upon which natural selection could act, and the origin of homochirality of amino acids and sugars. Conceptually, the genesis of the protein translation system posed a fundamental problem. Yet in the last decade significant progress has been made in all those areas, even though details are still sketchy and problems persist on many issues. Proposed reactions in the metabolism-first model, which assumes metabolism, not genes, at the origin of life, have also been rendered more promising by recent findings. Overall it can be said that puzzle pieces are starting to come together in such a way that the scientific assumption of a spontaneous origin of life from non-living matter finally has achieved plausibility on the level of experimental evidence.
While research in the field now appears vastly more promising than just a decade ago, the science on the origin of life is, compared to the science of biological evolution, still considerably underdeveloped in its explanatory power. As Richard Robinson notes (Robinson 2005): Give biologists a cell, and theyll give you the world. But beyond assuming the first cell must have somehow come into existence, how do biologists explain its emergence from the prebiotic world four billion years ago?
Indeed, it is one thing that we know all the chemical building materials of life, and that the functioning of life can be fully explained by their collaboration in an extremely complex system. Yet it is another thing entirely how, at the origin of life, they could have formed an initial organization by themselves step by step (via whatever intermediary processes and building blocks). At first glance, evolution from LUCA, a precursor of bacteria, to humans may seem childs play in comparison: it started from an already tremendously complex, entirely self-sufficient, biochemical machinery and bit by bit simply made it even more complex.
One cannot lose out of sight that the most elementary cells we currently know, which are not permanently dependent on host-metabolism, the bacterium Mycoplasma genitalium, have 482 protein-coding genes (most bacteria, such as E. coli, encode for more than 2000 different proteins), from which, according to the most thorough experimental study to date (Glass et al. 2006), the essential ones are 387. The likely most accurate hypothetical study (Gil et al. 2004) , puts the minimal number of genes at 206. All the proteins produced from these genes are involved in a maze of pathways of metabolism, replication, as well as building and maintenance of structure, which is of bewildering complexity.
In fact, how else than through such a minimum amount of complexity, could even a primitive cell have met the just mentioned basic demands? How could such a vastly complex network of more than 200 proteins have arisen by itself? One might ask: would it not have to have arisen at once? Yet evidence suggests that all this complexity may have evolved, step by step, from very simple beginnings.
2. Conditions for synthesis of organic molecules on
the early Earth
Essential to the spontaneous origin of life was the availability of organic molecules as building blocks. The famous prebiotic soup experiment by Stanley Miller (Miller 1953, Miller-Urey experiment) had shown that amino acids, the building blocks of proteins, arose among other small organic molecules spontaneously by reacting a mixture of methane, hydrogen, ammonia and water in a spark discharge apparatus. These conditions were assumed to simulate those on the primitive Earth. Already in 1922 Oparin had proposed that the early Earth had such a reducing atmosphere (in his classic The Origin of Life from 1936 he expanded on these ideas). Observations of Jupiter and Saturn had shown that they contained ammonia and methane, and large amounts of hydrogen were inferred to be present there as well (it is now known that hydrogen is the main atmospheric component of these planets). These reducing atmospheres of the giant planets were regarded as captured remnants of the solar nebula and the atmosphere of the early Earth was assumed by analogy to have been similar.
It was suggested that only in a reducing atmosphere like this, synthesis of organic molecules also sugars and organic bases, building blocks of nucleotides would have been possible in large amounts (Chyba and Sagan 1992).
Later research had cast doubt on the existence of a reducing atmosphere, and suggested a neutral atmosphere instead see also (Chyba 2005), the accompanying article to Tian et al. (see below).
However, new calculations indicate that hydrogen escaped from the early atmosphere at a much slower rate than previously thought, yielding an atmosphere where hydrogen was a major component (about 30 %) and which was therefore highly reducing (Tian et al. 2005 , see also press release). The authors measured the production of organic molecules through UV photolysis under those conditions, and conclude that at 1010kg/year it would have been orders of magnitude greater than the rate of either the synthesis of organic compounds in hydrothermal systems or the exogenous delivery of organic compounds to early Earth.
Another recent study supports an early reducing atmosphere as well. Chondrites are primitive material from the solar nebula and are generally believed to be the building blocks of the Earth and other rocky planets, asteroids and satellites. During and after planet formation, gases escape from the chondritic material due to high temperature and pressure. Systematic, detailed calculations on what these gases must have been show that they are mainly the highly reducing hydrogen, methane and ammonia the same gases as in the Miller-Urey-type experiments (Schaefer and Fegley 2007, see also press release). The composition of the gases varied with temperature only to a moderate extent, and was found to be largely independent of the actual pressure under which outgassing may have occurred, which appears to support robustness of the conclusions.
The authors mention that it
had been found that a reducing atmosphere of methane and ammonia is extremely
vulnerable to destruction by UV sunlight (Kuhn and
Atreya 1979, Kasting et al. 1983). They also point out, however,
that recent developments suggest that a
reducing atmosphere is more stable than previously believed:
1) It was found that hydrogen escape from the Earths atmosphere was less
efficient than previously thought (referring to the study by Tian et al. above).
2) Observations of the atmosphere of Titan, Saturns moon, which is composed
primarily of methane and nitrogen, show that photochemically produced
hydrocarbon aerosols form a haze layer in the upper atmosphere that protects
the lower atmosphere from photochemical destruction. Such a haze layer could
also have been produced on the early Earth from outgassed methane and ammonia
(Zahnle 1986, Sagan and Chyba
1997, Pavlov et al. 2000).
Yet even if the early Earths atmosphere would have been neutral rather than reducing, new data suggest that efficient amino acid synthesis would have been possible also under these circumstances. The group of Jeffrey Bada demonstrated that, contrary to previous reports, significant amounts of amino acids are produced from neutral gas mixtures under suitable conditions (Cleaves et al. 2008). Buffering with calcium carbonate (a common rock material) prevented lowering of the pH by produced nitrite and nitrate in the spark discharge apparatus used, dramatically increasing yield. Furthermore, addition of oxidation inhibitors prevented oxidative degradation of amino acids by nitrite and nitrate, increasing yield even further. Oxidation inhibitors such as ferrous ions may have been in excess over nitrite/nitrate on the early Earth (Walker and Brimblecombe 1985). The group of Bada also re-analyzed samples from Millers 1950s spark experiments simulating water vapor-rich volcanic eruptions. Such eruptions would have released reducing gases. In these samples amino acids were more varied than in the classical Miller experiment, and yields were comparable or even higher, indicating that even if the Earths atmosphere had been neutral, localized prebiotic synthesis could have been effective (Johnson et al. 2008, see also news article).
Of course, if life arose in deep-sea hydrothermal vents (see below), the composition of Earths early atmosphere would become largely irrelevant. To a certain extent, this also holds true for organic building blocks delivered to the earth by interplanetary dust particles and on carbonaceous meteorites.
In a reducing atmosphere, made likely with the findings discussed above, the concentration of organic compounds in the prebiotic ocean may have been relatively high (De Duve and Miller 1991, and references therein). Furthermore, locally a prebiotic soup might have been greatly concentrated by such simple processes as, for example, evaporation in puddles or shallow lakes, possibly with long-term wet/dry cycles. It should be kept in mind for evaluating all chemical scenarios that, due to its nature, the origin of life must have been a very local event; this is also important for the issue of the origin of homochirality of amino acids and sugars, see below.
It is now widely agreed that at the origin of life there was not the current DNA/(RNA)/protein system for gene information on one hand and catalysis, regulation, and structural function on the other. It would beg the question, what came first, protein or DNA? Protein catalysis without gene information, which allows it to be maintained and propagated, is not sufficient in the long term, and DNA gene information without catalysis, necessary for the function of life, would be useless as well.
Instead, it is assumed that RNA acted as a precursor of both protein and DNA, in the sense that it can serve both as catalyst (like protein enzymes) and as carrier of genetic information. Even in the modern cell ribozymes (catalytic RNAs) still play a vital, albeit limited, role. In the ribosome, the synthesis of the peptide chains of proteins from RNA code is accomplished by ribozymes. They also catalyze splicing of RNA.
The hypothesis that a so-called RNA World was involved in the early evolutionary stages of life is now an almost universally held view (Joyce 2002, Orgel 2004, The RNA World 2006). Could this RNA World have stood at the ultimate origin of life? This is currently still an open question. The RNA system may be too complex to have arisen without synthesis by a genetic precursor or prior enzyme-less metabolism (options discussed below). Yet while there are still substantial problems, there are now good leads for simple, spontaneous processes on the early Earth for both the synthesis of nucleotides and their concatenation to oligonucleotides.
For a long time the synthesis of RNA monomers under prebiotic conditions appeared to be a fundamental problem since the condensation of sugar (ribose) and nucleobase (purines and pyrimidines) does not work (Orgel, 2004). The prebiotic synthesis of purine ribonucleotides is still unclear, yet recently a breakthrough has been made with regard to the synthesis of pyrimidine ribonucleotide monomers (which incorporate cytosine and uracil). It now appears in principle to be solved, in a completely unexpected manner. The study by the group of John Sutherland (Powner et al. 2009) shows how nature could have spontaneously assembled pyrimidine ribonucleotide monomers from prebiotically plausible molecules through intermediates that contribute atoms to both the sugar and base portions of the ribonucleotides, thus avoiding a condensation step of sugar and base altogether (Fig.1). See also Nature News for the impact of these findings. While a good pathway for synthesis of purine ribonucleotides (incorporating adenine and guanine) still remains to be found, Jack Szostak argues in a comment accompanying the article (Szostak 2009) that it is precisely because this work opens up so many new directions for research that it will stand for years as one of the great advances in prebiotic chemistry.
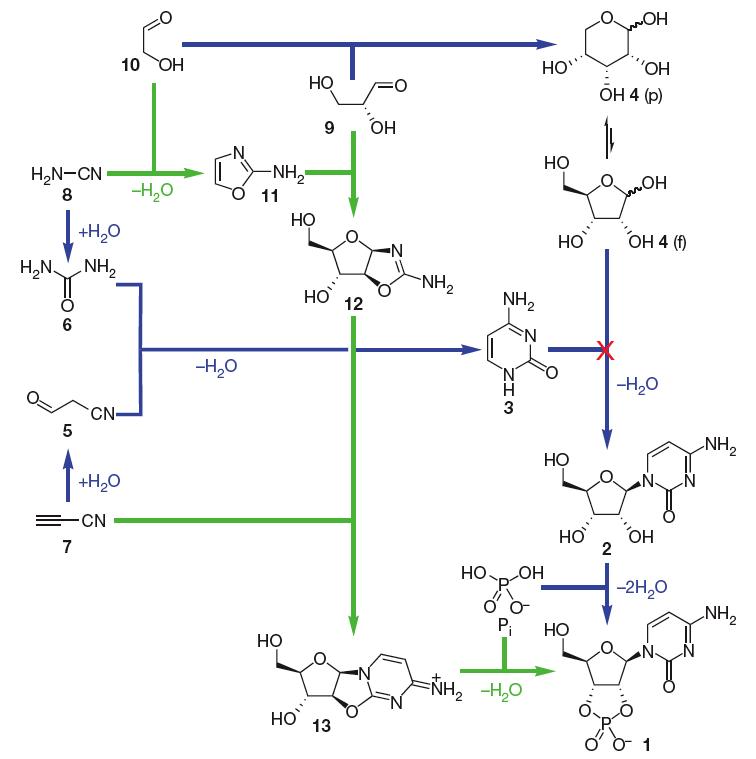
Figure 1. Pyrimidine ribonucleotide assembly options. Previously assumed synthesis of b-ribocytidine-2,3-cyclic phosphate 1 (blue; note the failure of the step in which cytosine 3 and ribose 4 are proposed to condense together) and the successful new synthesis described here (green). p, pyranose; f, furanose. From (Powner et al. 2009). Reprinted by permission from Macmillan Publishers Ltd.
(5 = Cyanoacetaldehyde, 6 = urea 6, cyanoacetylene 7, 8= cyanamide, 9 = glyceraldehyde, 10 = glycolaldehyde, 11 = 2-amino-oxazole, 12= pentose amino-oxazoline, arabinose derivative, 13 = anhydroarabinonucleoside)
With regard to the concatenation of nucleotides to oligonucleotides there is progress as well. The polymerization of chemically-activated RNA monomers can take place on the mineral surfaces of montmorillonite clay, generating polymer chains of up to 50-mers (Huang and Ferris 2006). The pyrimidine ribonucleotide monomers from the new synthesis (Powner et al. 2009) are also activated (they contain cyclic phosphate), which may allow for similar polymerization.
The group of David Deamer has shown that the synthesis of RNA-like polymers can even occur from non-activated mononucleotides within phospholipids vesicles, due to the chemical potential of fluctuating anhydrous and hydrated conditions, with heat providing activation energy during rehydration (Rajamani et al. 2008). Such conditions could have existed around hot springs on the prebiotic Earth. The lipids also provide a structurally organizing microenvironment that imposes order on mononucleotides. In this experimental setup, oligomers of up to 100 nucleotides can be formed non-enzymatically. It remains to be seen if prebiotically plausible fatty acid vesicles could have the same effect on RNA synthesis (with this a self-replicating RNA molecule would also have been pre-packaged for further evolution, cf. below). Effective polymerization of monomers that are activated might be aided by a structurally organizing microenvironment within vesicles as well.
However, while these reactions make RNA-like polymers they do not yet solve the problem of the stereospecific 3-5 concatenation of monomers (Orgel 2004), found in all living organisms. Both the lipid-assisted synthesis and polymerization on montmorillonite produce mixes of 2-5 and 3-5 bonds. Yet in the latter there is a preference towards 3-5 bonds (up to 74 %) which is promising. The authors of the study (Huang and Ferris 2006) argue that a higher proportion of 3-5 links could have resulted in the more rapid evolution to all 3-5 phosphodiester bonds during replication of oligomers, suggesting that perhaps complete 3-5 specificity of bonds within the first replicating RNA molecule may not have been required. This scenario may not be impossible, given that RNA usually forms more irregular secondary structures than DNA with its double helix, for which absolute stereospecificity of synthesis is mandatory. In the study, different kinds of chemical activation of RNA monomers yielded different preferences for 3-5 concatenation. It remains to be seen how specific for 3-5 bonds concatenation of monomers from activation with cyclic phosphate will be, the kind of activation reported in the new synthesis of pyrimidine ribonucleotide monomers (Powner et al. 2009).
Because of the difficulties of the synthesis of RNA nucleotides and oligonucleotides which, however, appear now considerably less severe than just a few years ago, given the new findings several alternatives to RNA as the first genetic system have been proposed, which may have preceded it. Peptide nucleic acid (PNA), threose nucleic acid (TNA) and peptides in which D- and L-amino acids alternate that incorporate standard nucleic-acid bases (ANAs) are discussed in (Orgel 2004). An even simpler system is glycerol nucleic acid (GNA, see Zhang et al. 2005). A highly interesting and chemically appealing, yet still untested idea is the PAH World (PAH = polycyclic aromatic hydrocarbons), developed by S. Platts and described in (Hazen 2005) and also at Wikipedia and at pahworld.com.
Other difficulties are that a ribozyme (catalytic RNA) that can copy itself completely has not yet been found so far, a 200 base ribozyme can copy around 20 bases of its sequence with high fidelity (Zaher and Unrau 2007), an improvement on (Johnston et al. 2001). A solution for the problem of copying a long ribozyme sequence has been proposed in Szostak et al. (2001); experiments will have to show if it is feasible. Also, the formidable problem of separating the double-stranded product of the copying reaction so as to permit a second round of copying would remain to be solved (Orgel 2004). Recent findings suggest that this problem may be addressed by thermal cycling, analogous to the polymerase chain reaction (PCR), which may have been a possibility under plausible prebiotic conditions (Mansy and Szostak 2008, Baaske et al. 2007).
4. Gradual build-up of complexity
Let us assume the plausible scenario that either RNA was directly synthesized, see above, so that out of a large pool of random RNAs a self-replicating RNA molecule could arise, or that such synthesis was accomplished by a precursor genetic/catalytic system (possibly on the surface of minerals, cf. Orgel 2004). Since fatty acids could have been available in the environment (Hanczyc et al. 2003, Orgel 2004), a primitive fatty acid membrane could have surrounded the first self-replicating RNA molecules (due to their molecular properties, fatty acids can form vesicles spontaneously); this would not have allowed passage of the RNA polymers so that they would have stayed together, but would have let the much smaller nucleotides through, fed in from spontaneous prebiotic synthesis or from a precursor genetic/catalytic system. Such a membrane would have had different characteristics of semi-permeability than modern lipid membranes, where a lot of molecule transfer is regulated through protein channels.
The group of Jack Szostak has performed extensive and plausible studies that these fatty acid vesicles as containers for RNA would have allowed growth and replication merely by physico-chemical mechanisms, until a more sophisticated membrane machinery, steered by the cell itself and more resembling what is found in current organisms, would have taken their place (Hanczyc et al. 2003, Chen et al. 2004, Hanczyc and Szostak 2004, Zhu and Szostak 2009).
While in earlier studies (Hanczyc et al. 2003, Hanczyc and Szostak 2004) more extreme conditions and sheer forces were required for vesicle division, also leading to the loss of a substantial fraction of vesicle contents, a new study (Zhu and Szostak 2009) shows a solution to these problems. It uses multilamellar vesicles (vesicles with several layers of lipid membrane) that form spontaneously by the rehydration of fatty acid films or by the acidification of a concentrated solution of fatty acid micelles. Once multilamellar vesicles are formed, spontaneous further incorporation of fatty acid micelles into them causes, via an unexpected mechanism, the formation of strongly elongated, thread-like vesicles. After subjection to mild shear forces these divide into several, again round, small daughter vesicles that preserve RNA contents well.
The group of Szostak also has demonstrated that nucleotides can pass through prebiotically plausible fatty-acid based vesicles and that non-enzymatic template copying of a model oligo dC DNA template can take place within them (Mansy et al. 2008), which, in connection with the studies of vesicle growth and division, reveals in principle how a heterotrophic protocell may have functioned (Fig. 2). Furthermore, they showed (Mansy and Szostak 2008) that prebiotically plausible model membranes are surprisingly thermostable, allowing them to tolerate at least short periods of temperatures of up to 100°C. Thermal cycling might have been possible near or within the surface of hydrothermal vents or hot springs (for thermal convection within hydrothermal pores, see also the below mentioned study by Baaske et al. 2007). This might solve the thorny issue of separating the double-stranded product of the copying reaction for further replication. Thermal cycling could have allowed for this separation of copying products (and increased nucleotide uptake, see Mansy and Szostak 2008) at high temperatures, and copying at lower ones, analogous to the polymerase chain reaction (PCR).
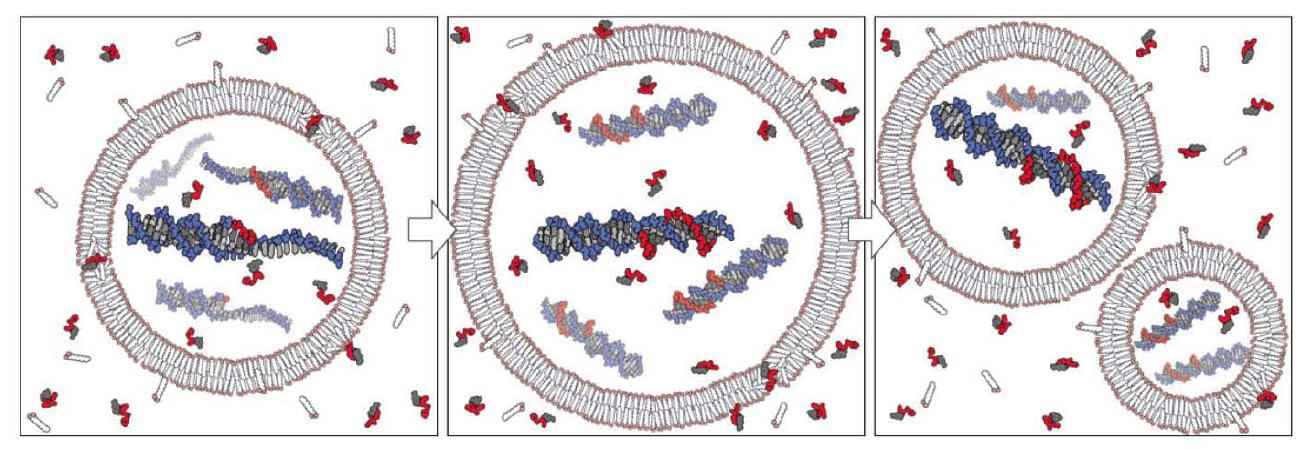
Figure 2. Conceptual model of a heterotrophic protocell. Growth of the protocell membrane results from the incorporation of environmentally supplied amphiphiles, whereas division may be driven by intrinsic or extrinsic physical forces. Externally supplied activated nucleotides permeate across the protocell membrane and act as substrates for the copying of internal templates. Complete template replication followed by random segregation of the replicated genetic material leads to the formation of daughter protocells. From (Mansy et al. 2008). Reprinted by permission from Macmillan Publishers Ltd.
Extrapolating from all the above data, inside fatty-acid vesicles the first self-replicating RNA molecule could have started copying itself. During copying, various things would have been possible. High-fidelity copies would have yielded the same self-replicating molecule. Copies with errors would mostly have resulted in RNA that was non-functional, but in a minority of cases, they could have yielded RNA that copied itself faster. It has been shown (Chen et al. 2004, see also news article) that RNA/vesicle systems that contain more genetic material (which would have resulted from faster RNA replication) develop more internal tension than neighboring vesicles that do not contain as much RNA, and draw membrane material from them. Importantly, this would have allowed for natural selection of vesicles by competition even in the absence of the ability to synthesize their own membrane components and therefore to directly control their own growth. Thus, for the first time, a system would have had the ability to undergo Darwinian evolution by natural selection acting on variation. This would have been a new and crucial emergent property arising at the transition from non-life to life.
A small portion of other copying errors (again, most substantial errors would probably have resulted in non-functional molecules, but those would have been filtered out by natural selection) could have led to RNA molecules with yet other, entirely different catalytic properties than the copying function. A new property could have allowed the RNA/vesicle system to even better compete for resources: just like in the case of the RNA molecules featuring the better copying function, the RNA would have evolved (1). The copy function of the parent molecules would probably have acted on these daughter molecules as well (like an RNA polymerase enzyme that copies any RNA). RNA/vesicle systems that had the altered RNA molecules with the new beneficial function, in addition to retaining the RNA with the copy function, would have been favored by natural selection. Finally, through reiteration of such processes, a series of new catalytic properties could, for example, have allowed the RNA pool within the vesicles to start making its own nucleotides. Would it then have been self-sufficient? To a certain degree, yes. Could this have been the first primitive cell? Why not?
It is just that in this scenario, the initial metabolism would have been much simpler than todays metabolism: Among others, energy metabolism could have been replaced by passage of activated building blocks for molecules from the outside environment into the vesicle (in a sense providing a preliminary substitute for modern-day ATP production, a possibility in view of the simple metabolism), and lipid metabolism, building of membrane structure and its regulation during replication would have been replaced by simple vesicles plainly obeying physico-chemical forces.
In other words: the cell would have depended more on the outside world, but for what it was doing, it was to a certain degree self-sufficient (todays organisms also need sustenance from the outside world of course, in the form of nutrients). On the other hand, the dependence of the cell on the outside world would also have been possible in a more immediate manner. A modern cell cannot, for example, use fatty acids from the outside in a way that they are directly incorporated as membrane elements.
Earlier we had asked: How could a complex network of more than 200 essential proteins, as it is found in todays most elementary cells, have arisen on its own?
The key to answering this question appears the combination of the above two attributes in the primitive cell: more direct dependence on the outside world than the modern cell, but also a greater ability to show such dependence by accepting molecular building blocks as such without having to convert nutrients into them. Starting with these characteristics and gradually moving on from there, evolution indeed could have eased the cellular system into more complexity.
Perhaps lipid synthesis, in a precursor form of modern synthesis, could have made the system more independent. The RNA system could have, bit by bit, invented protein synthesis as mentioned, the modern ribosomes still contain ribozymes (catalytic RNA) that catalyze the formation of peptide bonds which eventually result in proteins. In a compelling study (Wolf and Koonin 2007) the authors propose a stepwise model for the origin of the protein translation system, in which each step confers a distinct advantage onto an ensemble of co-evolving genetic elements. The goal of development of translation would not have been required, a foresight which evolution does not have. The initial cause for the emergence of translation would have been the ability of amino acids and peptides to stimulate reactions catalyzed by ribozymes (for peptides experimentally shown, see Robertson et al. 2004). Even if it will turn out that several steps in the evolution of translation probably have been different from the proposed model, the study clearly demonstrates that there is nothing in the emergence of the translation system that would represent a case of irreducible complexity, incapable of being subject to stepwise Darwinian evolution.
A recent study (Bokov and Steinberg 2009), based on analysis of the interdependence of elements of the macromolecule, shows a step-by-step evolution of the ribosome on the structural level, beginning from a very small core, the peptide transpeptidation centre. Further layers were added from the outside one by one, until the current complex macromolecule emerged. Each of these layers is structurally connected only to the respective previous one, and could be removed in the model without destroying the integrity of the inner layers (like the peeling of an onion), showing that there was no need for the entire molecule to emerge at once. This establishes the lack of irreducible complexity of the translational machinery from another perspective.
Finally, complex metabolism could have been achieved and the transition to the modern DNA/(RNA)/protein world. The dualism DNA/protein of course is a source of complexity in itself, one that is lacking in an RNA-only organism.
What about the difficult issue of a genome which holds all genes together? It might have been that in the first primitive cells RNAs were ligated by accident step by step, one by one, into forming a genome precursor and that each such step conferred an advantage in natural selection over competitor cells, since genes would not have been lost anymore during cell division, and replication would have been synchronized. Over time, an entire small RNA genome potentially could have organized itself in this manner, until mechanisms for internal expansion, like they are found in modern genomes, could have taken over, e.g. gene duplication and variation of the duplicated gene.
Certainly, reading of a string of ligated RNA as several single genes would have required that the primitive cellular mechanism would have come across a way to recognize beginning and end of a sequence. Promoter regions and start/stop codons, in the form they are used in protein-coding genes, would not have been present in a primitive RNA organism.
While lack of replication fidelity would have been an issue in primordial RNA genomes, analysis of experimental mutation studies on ribozymes indicates that an RNA genome may nonetheless have grown as large as 100 genes (Kun et al. 2005, Poole 2006). The RNA genome could, bit by bit, have been replaced by a DNA genome, a selectable advantage that primordial cells would have encountered by chance.
As for the universal genetic code: there is evidence that, until the last common ancestor, the genetic code was selected to a substantial extent for error minimization, thus is not arbitrary. Yet in part it is probably also frozen accident since the selection for this feature appears not to be maximal (Koonin and Novozhilov 2009).
In summary, based on available data a spontaneous origin of life as simple cells containing a single genetic polymer, upon which natural selection could act, is feasible. A gradual evolutionary transition from these to common cellular complexity would have been possible.
5. Minerals and the origin of life
Minerals most likely provide the clue to a number of issues regarding the origin of life. As discussed, they have been shown to catalyze polymerization of nucleotide-like molecules (Huang and Ferris 2006, Orgel 2004). They also have been demonstrated to allow for the prebiotic synthesis in sufficient purity of a nucleotide component that had proven elusive borate minerals stabilize ribose (Ricardo et al. 2004, see also news article; however, for a synthesis of ribonucleotide monomers without sugar intermediate, see above). Fatty acid vesicle formation is aided by minerals as well, and mineral particles could have wound up inside vesicles and there exhibited catalytic properties (Hanczyc et al. 2003, Hanczyc et al. 2007).
In a different putative scenario, minerals also play an interesting role. Instead of in an aqueous prebiotic soup on or near the surface of the earth, it has been hypothesized that life may have begun in the depths of the ocean, in the unique environment of deep-sea hydrothermal vents. It was shown that relevant organic molecules can be synthesized in reactions involving gases and minerals present at those sites, with CO and other small carbon-containing molecules as the carbon source. These reactions require the high temperatures, or a combination of the high temperatures and pressures, found in deep-sea hydrothermal vents. To name a few examples: Reaction of CH3SH with CO in the presence of FeS/NiS results in activated acetic acid, or a mixture of H2S, FeS/NiS, and CO (as the sole carbon source) yields acetic acid (Huber, Wächtershäuser 1997). Pyruvate is formed from CO in the presence of nonylthiol and FeS (Cody et. al 2000). Alpha-hydroxy and alpha-amino acids are synthesized from CO, cyano ligands and methylthio ligands upon catalysis by Fe-Ni precipitates, in the presence of calcium or magnesium hydroxide (Huber, Wächtershäuser 2006). For criticism of the conditions in the latter study, however, and response see (Bada et al. 2007).
Furthermore, in high-pressure, high-temperature water as found in deep-sea hydrothermal vents, organic molecules, once formed, show a level of (albeit not always particularly specific) chemical reactivity that is usually observed in normal aqueous environments only upon speeding-up of reaction rates by enzymes (see for example the review Hazen et al. 2002). For the physico-chemical properties of high-pressure, high-temperature water, see Basset M-P 2003 (water behaves more like an apolar organic solvent under these conditions). Catalysis by minerals, such as those present in deep-sea hydrothermal vents, further enhances reactions of organic molecules in such an aqueous environment (2). Degradation of synthesized organic molecules under these high-temperature and high-pressure conditions may be prevented by minerals as well at least this has been shown for amino acids (see Hazen et al. 2002). Fatty acids, as a source of membrane-forming material, might have been synthesized in hydrothermal vents too (Orgel 2004).
In addition there are, also just warm to moderately hot, hydrothermal vents that have semi-permeable microenvironments of cell-like dimensions (mimicking a lipid membrane), which could retain molecules at high concentrations. This would provide a possible solution to the concentration problem (Russell and Martin 2004, Robinson 2005). However, it remains to be seen in which way such immobile compartments would have allowed for natural selection, which is possible in the case of fatty acid vesicles by competition for membrane material (Chen et al. 2004) and other resources, as discussed here. Koonin and Martin (2005) propose that, if the mineral micro-compartments had a certain porosity, more competitive evolutionary units could have spread over others by preferentially occupying new, empty compartments that should form continuously by sulfide precipitation at hydrothermal vents. This would have been a progenitor of cell division. The alternative which, in contrast to this interesting yet still hypothetical model, already enjoys extensive experimental support would of course have been division of fatty acid vesicles containing genetic polymers by simple physico-chemical forces, see above. The authors also argue (cf. Martin and Russell 2003) that mineral micro-compartments instead of lipids might have served as cell membrane even to such an advanced organism as LUCA, since in their view this would allow for the simplest explanation why the descendents of LUCA, archebacteria and eubacteria, have very different lipid membranes these would have arisen only later.
Hydrothermal vents may offer yet another solution to the concentration problem for key components in the origin of life. RNA monomers and oligonucleotides may have been greatly accumulated in hydrothermal pore systems due to a thermal gradient across the pore (Baaske et al. 2007, see also the accompanying comment, Koonin 2007). Up to 1000fold concentration of nucleotides is achieved in a single pore, and more than 108 fold in concatenated pores (these pores are up to millimeter-sized, thus much larger than the above micro-compartments). This accumulation is not dependent on adsorbing surfaces that might be restrictive to sequences of reactions. The group of Jack Szostak (Budin et al. 2009) showed that also fatty acids are greatly concentrated in similar systems. When concentrating dilute solutions of both DNA and fatty acids, the self-assembly of large vesicles containing encapsulated DNA occurs in regions of the capillaries where the critical aggregate concentration of the fatty acid is exceeded.
All these concentration scenarios may not just work in deep-sea hydrothermal vents, but also in hydrothermal systems that are found in shallow waters in the periphery of volcanoes. The thermal convection within hydrothermal pores (Baaske et al. 2007) would also have allowed for thermal cycling to separate the double-stranded product of RNA copying reactions.
Chemistry in these
environments may open up new possibilities for the synthesis of a genetic polymer
preceding RNA, or of RNA itself, required in the gene-first scenario.
6. Specificity of chemical reactions and the metabolism-first scenario
Some extend the above findings from deep-sea hydrothermal vents to a variant of the metabolism-first scenario as opposed to the gene-first scenario described above, a hypothesis that claims that complex metabolic cycles could have self-organized, independent of a genetic system which is capable of providing polymer catalysts. According to this hypothesis, only via such a stable metabolism the synthesis of nucleotides and oligonucleotides, necessary for the inception of the RNA world, would have been possible yet due to the here discussed recent findings spontaneous prebiotic RNA synthesis is now becoming far more likely than when these metabolism-first scenarios were originally proposed.
In the same publications in which Günter Wächtershäuser had predicted the synthesis of organic molecules under conditions as found in hydrothermal vents, predictions that have been experimentally confirmed (see above), he also introduced a metabolic model (Wächtershäuser 1988, Wächtershäuser 1990). This inventive and detailed hypothesis integrates in an impressive manner a multitude of observations in the field of chemistry. It suggests that the beginning of life was what has been termed a flat life, an elaborate two-dimensional metabolism on mineral surfaces of deep-sea hydrothermal vents; this also addresses the dilution problem by concentrating all chemistry on the surface. Central to this and other metabolism-first hypotheses is the reductive citric acid cycle (reverse Krebs cycle) which provides a core mechanism of useful biomolecules from CO2 (Morowitz et al. 2000, Smith and Morowitz 2004).
However, the unfolding of this and related scenarios (e.g. Smith and Morowitz 2004) would require a surprising lack of side reactions (while a residual small level of side reactions might favor evolutionary development). It is not easy to see how the extraordinarily high specificity of chemical reactions, required for complex sequences of reactions and metabolism for the functioning of life , would in general be possible without catalytic polymers featuring a three-dimensional substrate pocket. These are only provided by a gene-first scenario. Leslie Orgel (Orgel 1998) comments on the issue as follows (for an extended critique, see Orgel 2000):
There is no agreement on the extent to which metabolism could develop independently of a genetic material. In my opinion, there is no basis in known chemistry for the belief that long sequences of reactions can organize spontaneously and every reason to believe that they cannot. The problem of achieving sufficient specificity, whether in aqueous solution or on the surface of a mineral (3), is so severe that the chance of closing a cycle of reactions as complex as the reverse citric acid cycle, for example, is negligible. The same, I believe, is true for simpler cycles involving small molecules that might be relevant to the origins of life and also for peptide-based cycles.
Thus it appears reasonable to assume that the development of metabolic cycles and pathways would have required genetic/catalytic polymers even though, obviously, opinions are divided on the issue.
Stuart Kauffmans large autocatalytic sets (Kauffman 1993) may be far too optimistic for the same reason. Autocatalytic sets may have a better chance of being realized when they are small.
Newer developments, however, may be seen to raise the possibility that the metabolism-first scenario is realistic after all. It was found (Zhang and Martin 2006) that three of the five reductive steps of the reverse Krebs cycle (Fig. 3) could be driven by ZnS particles (providing reducing power of conduction-band electrons and believed to have been prevalent in the waters of the early Earth) under the influence of UV light. The conversions of oxalacetate to malate, and of fumarate to succinate proceeded at astonishing yields (75 % and 95 %, respectively), which may be close to sufficient. The conversion of succinate to oxoglutarate also proceeded, albeit at a low rate of 2.5 %. The authors suggest that a more complex mineral assemblage than ZnS alone may drive the entire suite of reactions.
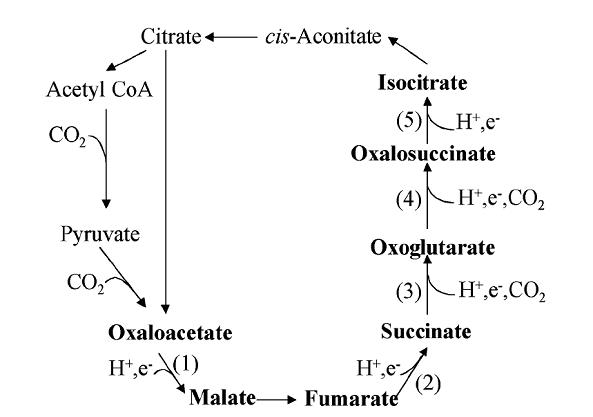
Figure 3. Reverse Krebs cycle. Highlighted are the five reduction reactions (labeled 1-5). From (Zhang and Martin 2006). Reprinted by permission. Copyright 2006 American Chemical Society.
Of high interest are also recent findings that have been reported (Robinson 2005) to give hope to adherents of the metabolism-first model. It has been shown (Cordova et al. 2005a) that such simple organic molecules as single amino acids can catalyze the stereospecific synthesis of sugars from simple starting materials with enzyme-like specificity, albeit only in organic solvents. For example, either L- or D-enantiomers of certain amino acids (with serine among those, see below) can trigger formation of a certain type of sugar not just with excellent chemoselectivity (i.e. avoiding unwanted side reactions), but also the formation of one out of 16 possible enantiomers of this sugar with approx. 99 % stereospecificity. These reactions are based on the principle of asymmetric organocatalysis discovered in the 1970s (Eder et al. 1971, Hajos and Parrish 1974).
Even more promising are the findings in a follow-up study (Cordova et al. 2006). Here the authors report that small peptides (mainly dipeptides tested, or peptides with no more than five amino acids) (4) and amino acid tetrazoles can catalyze aldol reactions, some of them yielding sugars, with great stereospecificity in water, not just in organic solvents.
Could highly specific catalysis by amino acids or other small organic molecules (5) more generally, i.e. not just in this particular reaction, substitute to a certain extent for the three-dimensional substrate pockets of catalytic polymers? Could it close metabolic cycles?
These speculative possibilities, involving specific catalysis by organic molecules, would come closer to the metabolism-first scenario that was proposed by Christian De Duve (De Duve 1995). He assumes a protometabolism involving a thioester world providing also energy for molecular reactions which would have become the basis for the emergence of the RNA world (this scenario was suggested for the prebiotic soup, not for chemistry in hydrothermal vents). However, De Duve bases his catalysis on multimers derived from thioesters, structurally similar to the [first] small catalytic proteins. It is hard to envision how such relatively large complex catalytic units could have constantly formed with high reproducibility in a spontaneous manner. Such reproducibility would have been required for establishing and maintaining a stable metabolism, or otherwise complex sequences of reactions of course, it poses no problem for genetic polymers, as provided by the gene-first scenario. Yet reproducibility (and abundance) of gene-less synthesis of putative small molecule catalysts would likely have been a much lesser issue.
Could the unique high-pressure, high-temperature aqueous environment of deep-sea hydrothermal vents, which produces drastic changes in the reactivity of organic compounds (see above), also cause small organic molecules to act as specific catalysts that would not perform this function in normal aqueous solution?
Finally, to solve the main
puzzle of the gene-first model: could catalysis by small organic molecules
even be involved in synthesis of nucleotides and oligonucleotides in the form
of the right stereoisomers a far more complex chemistry than just the
synthesis of sugars in the absence of precursor genetic/catalytic polymers?
Future research may inform us about all these issues.
It should be noted that the dependence on UV light
for the described reactions of the reductive citric acid cycle (Zhang and Martin 2006), possibly in conjunction with a requirement for moderate
temperatures under which these reactions were reported to proceed, would confine this chemistry to the Earths surface. As
Leslie Orgel points out (Orgel 2006), while historically metabolism-first
proponents mostly have argued for synthesis in deep-sea hydrothermal vents and
gene-first proponents for a prebiotic soup on the Earths surface, neither
connection may be an a priori prerequisite for the respective model
metabolism without genes could have developed on the Earths surface, or genes
without prior metabolism in hydrothermal vents.
On the other hand, while both these reactions of the reductive citric acid cycle and the new synthesis of activated pyrimidine ribonucleotide monomers (Powner et al. 2009) are reported to require UV light, reactions seemingly confined to one location may be shown, as knowledge advances, to also proceed in other locations under different conditions. Synthesis of RNA monomers in hydrothermal systems could be an attractive scenario since, as discussed above, hydrothermal pores would allow for great accumulation of RNA monomers and oligomers by thermal gradients across them (Baaske et al. 2007).
7. Origin of the homochirality of amino acids and
sugars
Life almost exclusively synthesizes L-amino acids and D-sugars. A key question related to the origin of life is the emergence of this homochirality (one-handedness). Homochirality is essential for the functioning of proteins as amino acid polymers, and for the structure of DNA and RNA, which requires incorporation of D-sugars.
How did this homochirality of amino acids and sugars arise? It is a question that has puzzled origin-of-life researchers for decades, yet a series of recent findings addresses the problem astonishingly well.
Enantiomers (L- or D-forms) of amino acids can be strongly enriched by two steps:
1. An initial imbalance of enantiomeric forms of an amino acid
How could a greater presence of one enantiomeric form of an amino acid over the other (enantiomeric excess, abbreviated: ee), be it ever so slight, have arisen at all? After all, typical synthesis of an amino acid in the laboratory results in an exact 1:1 ratio of the L and D enantiomer a racemic mixture.
One possible source are meteorites. On the Murchison meteorite, a well-studied example, the L-form of some of the amino acids found has been reported to be present in up to 9 % ee (Cronin and Pizzarello 1997). These ees may have been induced by circularly polarized UV light (Bailey et al. 1998). A recent study (Glavin and Dworkin 2009) reports ees of L-isovaline of more than 18 % on the Murchison meteorite, and postulates as cause aqueous alteration (a process in which changes to minerals occur influenced by water). Amino acids such as isovaline, which is not common in the terrestial biosphere, may have transferred chirality to biogenic compounds as catalysts in chemical reactions (Pizzarello and Weber 2004).
Yet while chemical evolution of early life could very well have built upon meteoritic material, sources of slight or even pronounced enantiomeric excesses of amino acids could have arisen in numerous places on the prebiotic earth.
Robert Hazen and colleagues found (Hazen et al. 2001) that crystals of the common rock-forming mineral calcite (CaCO3) can preferably adsorb D- or L-forms of aspartic acid (and in preliminary experiments, D- or L-forms of alanine as well) depending on the chirality of the crystal surface. Average ee was a few percentage points. In a similar manner, adsorption of 3-carboxy adipic acid, a prebiotically relevant molecule, to calcite or feldspar crystals produced ees of up to 10 % (Castro-Puyana et al. 2008).
Even though such slight local enantiomeric excesses may be enough to trigger events that can strongly amplify them (see below), another study found much larger preferences of adsorption of amino acids to mineral surfaces (Wedyan and Preston 2005). While in their study quartz, kaolin and montmorillonite showed slight preferences for adsorption of enantiomers, ordinary sediments from estuaries exhibited strong selectivity. Typical D/L ratios were hugely different from 1, reaching at their most extreme up to 100 for serine. Sediments were ashed in order to remove organic matter which could introduce chiral bias. The authors are cautious: The possibility that the ashing process actually etches and activates the mineral surface as it burns off natural (chiral?) organic matter cannot be discounted. However, they also note that it is remarkable that such strong selectivity should occur at all.
An entirely different, attractive mechanism to achieve enantiomeric excess has been reported (Kojo et al. 2004), which involves formation of a solid phase of amino acid crystals. This could well have happened in a prebiotic landscape by cooling down of a warm concentrated aqueous solution of amino acids or simply by slow evaporation of a solution. Most racemic amino acids form crystals that are also racemic. However, when racemic D, L-asparagine forms crystals they are not racemic, but show varying degrees of excess of either the L-enantiomer or the D-enantiomer. When other amino acids are present, either their L- or their D-form preferentially co-crystallizes with the enantiomeric form (L or D) of asparagine that confers an excess during formation of the particular crystal at hand.
The behaviour of amino acids in the study showed great consistency. If there was a bias towards L-asparagine in one crystal, practically all the other 12 co-crystallized amino acids showed a bias towards the L-form as well. The degree of bias towards L-asparagine varied between crystals; when the bias was more pronounced, the bias towards the L-form in the other amino acids was also more pronounced. The ees observed could be very high. If in yet another crystal there was a bias towards the D-form of asparagine, the other co-crystallized amino acids exhibited a bias towards their D-enantiomer as well.
Of course, the overall enantiomeric balance of the entire amino acid mixture (the sum of all crystals and the liquid phase above them) was still racemic. However, if, these kinds of crystals were formed from a solution on the prebiotic earth, and some of the crystals were then physically separated from others which could have happened by many ordinary processes and later redissolved, the amino acids in solution would automatically have exhibited an ee. Depending on the particular crystal where the solution derived from, this ee could have been high.
Another way to achieve enantiomeric excess within a racemate are special conditions of crystallization far from equilibrium. Normally aspartic and glutamic acids form racemic crystals. Yet when they are subjected to capillary rise through a porous material (such as a brick partially immersed in a solution), which is accompanied by supersaturation due to evaporation of solution through the porous structure, pure L- or D-crystals are formed (Viedma 2001). Similar conditions may have been present on the early Earth in sedimentary environments, like in a playa or sand bar.
2. Enhancement of enantiomeric excess by solid phase-liquid phase equilibria
Studies have shown that, once an initial excess of one enantiomer in a mixture of amino acids exists, even if it is just very slight, it can have an enormous effect. This effect can occur when solid and dissolved amino acids from such a mixture coexist in equilibrium, i.e. when crystals form upon, for example, limited evaporation of a solution.
A detailed study was
performed by the group of Donna Blackmond (Klussmann
et al. 2006). When a
mixture of L- and D- enantiomers of an amino acid shows an excess of one
enantiomer, in most cases an equilibrium of solid and dissolved amino acid will
consist of the following two or three components:
a) racemic crystals (crystals with a 1:1 ratio of the L- and D-enantiomers, no
enantiomeric excess either way)
b) pure crystals of the enantiomer in excess
(Whether just one of these two solid phases is present, or both coexist, will
depend on the overall ee.)
c) an amino acid solution in equilibrium with the solid phase(s) which also
exhibits a certain ee.
Yet as it turns out, for several amino acids this ee in solution is much higher than the overall ee of the total mixture (the solid material, dominated by racemic crystals, thus shows correspondingly less ee). At the most extreme, serine provides an almost enantiopure solution (> 99 % ee) in water from a nearly racemic sample (only about 1 % ee) under solid-liquid equilibrium conditions.
A smaller study, independently conducted around the same time, reports similar findings (Breslow, Levine 2006). Slow evaporation of an aqueous solution of phenylalanine at just 1 % ee of the L-enantiomer led to a solution of this amino acid with 40 % ee of the L-enantiomer above solid material. If, in turn, such a solution was allowed to evaporate, the resulting solution in equilibrium with the solid material had a 90 % ee.
In a more recent study, the Blackmond group (Klussmann et al. 2007) expanded the concept to mixtures of amino acids with other compounds, which can co-crystallize with the amino acids. They showed that, by influencing solubility, in some cases these compounds strongly influenced the ee in solution under solid-liquid equilibrium conditions. For example, under those conditions the ee of valine was raised from 47 % to up to 99 % in the presence of fumaric acid. Note that prebiotic plausibility is enhanced in this scenario, since it employs compound mixtures rather than pure components.
Another way of increasing ee does not employ enrichment of ee in solution but in the solid phase of a solid-liquid equilibrium. A curious mechanism of crystal growth, which involves attrition by grinding, allows for the emergence of a single chiral state in the solid phase from an initial small imbalance in crystal composition as low as 2-3 % ee (Noorduin et al. 2008). It does require racemization of the compound in solution, however, and has been applied to an amino acid derivative that undergoes this racemization upon addition of base. A theoretical explanation that differs from the one given in the study is discussed in a commentary on the findings (McBride and Tully 2008). In a follow-up study the findings were extended to an amino acid proper, aspartic acid (Viedma et al. 2008). Racemization in solution was accomplished with catalytic salicylaldehyde in acidic medium, and up to 99 % ee in crystals was obtained from an initial imbalance of less than 10 % ee. Heating could substitute for mechanical grinding.
In the studies by Klussmann et al. 2006 and by Breslow, Levine 2006 the findings are discussed also in terms of amino acid catalysis of aldol reactions, which include sugar synthesis and might result in enantiopure D-sugar, see also the findings by Cordova et al. 2005a and Cordova et al. 2006 reviewed above. Yet when the new pathway of synthesis for activated pyrimidine ribonucleotide monomers (Powner et al. 2009) is considered, which sidesteps base and sugar molecules and their condensation, but instead produces the final molecules via intermediates which contribute atoms to both the sugar and base portions of the ribonucleotides, the relevance of amino acid catalysis of D-sugar production or any other potential involvement of enantiomerically enriched amino acids in nucleotide synthesis is not clear. Since glyceraldehyde is one the starting molecules in the new synthesis (Fig. 1), chirality in RNA could have arisen by an input of homochiral glyceraldehyde, the origin of which might have resulted from processes similar to the ones discussed for amino acids.
In any case, it has also been shown that once an enantiomeric excess of amino acids was present, reactions catalyzed by them could have enhanced ee even further. It was shown that in the catalysis of sugar synthesis by amino acids a significant amplification of ee can occur (Cordova et al. 2005b). For example, a reaction mediated by the amino acid proline with an ee as low as 40 % still yielded almost enantiopure hexose sugar. The reaction catalyzed by proline at 10 % ee furnished the sugar with 33 % ee.
*****
In summary, a plausible
scenario for the emergence of homochirality on the prebiotic earth can be
envisioned from all the findings described:
1. Origin of local enantiomeric excesses slight or pronounced of amino
acids
2. In case these ees are just slight, a tremendous amplification by solid
phase-liquid phase equilibria; these can also enhance already high ees
As we have seen in the example of sugar synthesis, catalysis by amino acids could have played a role in the emergence of other chiral products relevant to the origin of life, and chiral amplification could have taken place in the process as well.
[If specific enantiomers of small peptides were the catalysts (cf. above), they could have arisen by conjugation of amino acids after enantiomeric enrichment.]
*****
Once homochirality arises in some biomolecules, it can aparently force corresponding homochirality onto others. We have seen this with amino acid catalysis, but it could also hold for the relationship between RNA and amino acids. Once D-sugars were present in RNA, the step towards L-amino acids in proteins might have been an automatic one. Protein synthesis requires the aminoacylation of RNA, and it is in this step that L-amino acids could have been selected for, as an elegant study shows (Tamura, Schimmel 2004).
In the study, an RNA minihelix was used that recapitulates the domain within transfer RNAs which harbors the amino acid attachment site. Such RNA minihelices are thought to be the progenitors of modern transfer RNAs, which present amino acids bound to them to the ribosomal machinery that makes proteins. The authors showed that the RNA minihelix was aminoacylated by activated amino acids with a clear preference for L- as opposed to D-amino acids. A mirror-image RNA system showed the opposite selectivity.
Three amino acids alanine, leucine and phenylalanine were tested, and the observed selectivity was 4-fold. The authors point out in a subsequent publication, which studies the mechanism (Tamura, Schimmel 2006), that such a 4-fold effect, repeated under selective pressure many times, can lead to an overwhelming preference for an L-amino acid in a biological system.
On the other hand, peptide chains could also have acquired chirality independent of RNA, see for example Saghathelian et al. 2001, see also press release, Hitz and Luisi 2004, Weissbuch et al. 2004, Plankensteiner et al. 2005. Of course, any such mechanisms in peptides could have been aided as well by an enantiomeric excess of amino acids as achieved in the above described scenarios.
Whatever the precise sequence of events at the origin of life may have been, the cumulative strength of all of the above data indicates that the mystery of the origin of lifes chirality can now well be explained after all.
*****
The issue of chirality, among others, has been touted by creationists as a "huge problem" for the concept of an origin of life by natural causes. Allegedly, only a miraculous intervention by God could have solved the problem. Yet the above findings are a typical example for why the God-of-the-gaps concept does not work: science rapidly closes the gaps that previously might have been thought to be reserved for miraculous intervention.
This is exactly what should be expected if either the material world is all there is, or if the world was created by a God who, as primary cause, chose to create through secondary causes precisely those natural causes that science studies. In fact, creationists should seriously ask themselves if their concept of God is not a belittling one: the Intelligent Designer as "tinkerer" who is forced to break his own created laws of nature once in a while because they are insufficient to achieve certain stages in the development of the material world. From a theistic philosophical perspective, the actual findings of science suggest a much grander idea of God: the Designer who laid out an elegant and self-sufficient set of laws of nature that accomplish the unfolding of his creation by inducing self-organization of the material world. This idea is easily compatible with the concept of God of many mainstream religions, including most Christian ones.
Hypercycles, as an overarching organization of autocatalytic sets, have been proposed as a model for the origin of life (Eigen and Schuster 1977) and are explained at Principia Cybernetica Web. However, complex hypercycles only exist as computer simulations. More than thirty years after the introduction of the hypothesis, there is no experimental evidence whatsoever for complex, possibly prebiotic hypercycles. This makes them still nothing more than mere speculation and, while the hypothesis has been a recurring theme in the scientific literature about the origin of life, it is not frequently mentioned anymore in recent publications in the field.
So far only one, very simple, hypercycle has been generated more than 10 years ago in the laboratory with two self-replicating peptides by the group of Ghadiri (Lee et al. 1997). However, these authors posted a correction (Lee et al. 1998) where they state: Although the kinetic data suggest the intermediary of higher-order species in the autocatalytic processes, the present system should not be referred to as an example of a minimal hypercycle in the absence of direct experimental evidence for the auto-catalytic cross-coupling between replicators.
There is a single report on a naturally occurring hypercycle (Eigen et al. 1991). But this example, relating to the infection cycle of an RNA bacteriophage, obviously is not prebiotic the cycle evolved on the complexity of living beings as template.
Even though recent, exciting research has provided plausible scenarios for the origin of life and has answered many questions, it is clear that a lot of research remains to be done, since much of the origin-of-life scenarios is still hypothesis. Experimental models are needed that are both realistic and of some appreciable complexity. Were it possible, for example, to show that a primitive RNA organism could be built in the laboratory (Szostak et al. 2001), it would be a significant step forward. For this, see Carl Zimmers article; there also the hope is expressed that evolution of such an organism might be observable on the lab bench.
(1) Outside a cellular context, RNA evolution in the laboratory is routine now RNA engineering according to mechanisms somewhat resembling evolution has been shown to produce many RNA catalysts (see e.g. Robertson et al. 2004). Certainly, all the sophisticated procedures used in the laboratory would not have been available to an ancient precursor of a cell. On the other hand, nothing in the test tube quite resembles the possibilities that would arise from the competition of replicating cells.
(2) The hypothesis that life might have originated in hydrothermal vents appears, to some extent, attractive also due to the fact that metal sulfides, found in those locations and capable of catalysis of simple organic reactions, are still found in the catalytic centers of central metabolic enzymes such as ferredoxin, succinate dehydrogenase and bacterial acetyl‑CoA synthase possibly hinting at evolutionary preservation of primitive metal sulfide catalysis.
(3) Wächtershäuser presents interesting theoretical arguments why surface metabolism, as opposed to reactions in solution, should address reaction specificity in a sufficient manner, yet his viewpoint has also been criticized by Christian De Duve and Stanley Miller in a joint article (De Duve, Miller 1991). Experimental evidence for the specificity of a sequence of reactions strictly confined to a mineral surface (not just catalyzed by it, which would allow for diffusion of intermediary products away from and back to catalytic surfaces) would be hard to obtain, and also Wächtershäusers own experimental publications, as well as those of others testing his metabolism-first scenario, thus far do not really address this issue so central to his hypothesis, but instead concentrate on other, simpler aspects of his model.
(4) Several examples of how
prebiotic oligomerization of amino acids to small peptides could have occurred:
a) Via the salt-induced peptide formation
reaction (SIPF), possibly in puddles, tidal pools or lagunas (Schwendinger, Rode 1989)
b) By volcanic gas (Leman et al. 2004)
c) In deep-sea hydrothermal vents, by activation with carbon monoxide
(CO) in the presence of minerals (Huber et al. 2003)
(5) Highly specific primarily in terms of chemoselectivity; in terms of stereospecificity only where applicable. This specific catalysis could also take place in conjunction with complex formation on mineral-surfaces as found in deep-sea hydrothermal vents, or organic molecules might form complexes with metal solubilized in this high-temperature, high-pressure environment (cf. Cody et al. 2000).
Baaske P, Weinert FM, Duhr S, Lemke KH, Russell MJ, Braun D (2007) Extreme accumulation of nucleotides in simulated hydrothermal pore systems. Proc. Natl. Acad. Sci. USA 104: 9346-9351 [Pubmed] [Free Full Text]
Bada JL, Fegley B Jr, Miller SL, Lazcano A, Cleaves HJ, Hazen RM, Chalmers J (2007) Debating evidence for the origin of life on Earth. Science 315:937-939 [Pubmed]
Bailey J, Chrysostomou A, Hough JH, Gledhill TM, McCall A, Clark S, Menard F, Tamura M (1998) Circular polarization in star-formation regions: implications for biomolecular homochirality. Science 281: 672-674 [Pubmed]
Basset M-P (2003) Is high-pressure water the cradle of life? JPCM 15: 1353-1361 [Abstract, free full text available there]
Bokov K, Steinberg SV (2009) A hierarchical model for evolution of 23S ribosomal RNA. Nature 457: 977-980 [Pubmed]
Breslow R, Levine SM (2006) Amplification of enantiomeric concentrations under credible prebiotic conditions. Proc. Natl. Acad. Sci. USA 103:12979-12980 [Pubmed] [Free Full Text]
Budin I, Bruckner RJ, Szostak JW (2009) Formation of protocell-like vesicles in a thermal diffusion column. J. Am. Chem. Soc. 131: 9628-9629 [Pubmed] [Full article on the Szostak lab publication page]
Castro-Puyana M, Salgado A, Hazen RM, Crego AL, Alegre ML (2008) The first contribution of capillary electrophoresis to the study of abiotic origins of homochirality: Investigation of the enantioselective adsorption of 3-carboxy adipic acid on minerals. Electrophoresis 29:1548-1555 [Pubmed]
Chen IA, Roberts RW, Szostak JW (2004) The emergence of competition between model protocells. Science 305:1474-1476 [Pubmed] [Full article on the Szostak lab publication page]
Chyba C, Sagan C (1992) Endogenous production, exogenous delivery and impact-shock synthesis of organic molecules: an inventory for the origins of life. Nature 355:125-132 [Pubmed]
Chyba CF (2005) Rethinking Earth's early atmosphere. Science 308: 962-963 [Pubmed]
Cleaves HJ, Chalmers JH, Lazcano A, Miller SL, Bada JL (2008) A reassessment of prebiotic organic synthesis in neutral planetary atmospheres. Orig. Life Evol. Biosph. 38:105-115 [Pubmed]
Cody GD, Boctor NZ, Filley TR, Hazen RM, Scott JH, Sharma A, Yoder HS Jr. (2000) Primordial carbonylated iron-sulfur compounds and the synthesis of pyruvate. Science 289:1337-1340 [Pubmed]
Cordova A, Ibrahem I, Casas J, Sunden H, Engquist M, Reyes E (2005a) Amino acid catalyzed neogenesis of carbohydrates: a plausible ancient transformation. Chemistry 11: 4772-4784 [Pubmed]
Cordova A, Engquist M, Ibrahem I, Casas J, Sunden H. (2005b) Plausible origins of homochirality in the amino acid catalyzed neogenesis of carbohydrates. Chem. Commun. (Camb) (2005): 2047-2049 [Pubmed]
Cordova A, Zou W, Dziedzic P, Ibrahem I, Reyes E, Xu Y (2006) Direct asymmetric intermolecular aldol reactions catalyzed by amino acids and small peptides. Chemistry 12: 5383-5397 [Pubmed]
Cronin JR, Pizzarello S (1997) Enantiomeric excesses in meteoritic amino acids. Science 275: 951-955 [Pubmed]
De Duve C, Miller SL (1991) Two-dimensional life? Proc. Natl. Acad. Sci. USA 88: 10014-10017 [Pubmed] [Free Full Text]
De Duve, C (1995) The beginnings of life on earth. American Scientist 83: 428-437 [Free Full Text]
Eder U, Sauer G, Wiechert R (1971) New Type of Asymmetric Cyclization to Optically Active Steroid CD Partial Structures. Angew. Chem. Int. Ed. Engl. 10:496-497
Eigen M, Schuster P (1977) The hypercycle. A principle of natural self-organization. Part A: Emergence of the hypercycle. Naturwissenschaften 64: 541-565
Eigen M, Biebricher CK, Gebinoga M, Gardiner WC (1991) The hypercycle. Coupling of RNA and protein biosynthesis in the infection cycle of an RNA bacteriophage. Biochemistry 30:11005-11008
Gil R, Silva FJ, Pereto J, Moya A (2004) Determination of the core of a minimal bacterial gene set. Microbiol. Mol. Biol. Rev. 68: 518-537 [Pubmed] [Free Full Text]
Glass JI, Assad-Garcia N, Alperovich N, Yooseph S, Lewis MR, Maruf M, Hutchison CA 3rd, Smith HO, Venter JC (2006) Essential genes of a minimal bacterium. Proc. Natl. Acad. Sci. USA 103: 425-430 [Pubmed] [Free Full Text]
Glavin DP, Dworkin JP (2009) Enrichment of the amino acid L-isovaline by aqueous alteration on CI and CM meteorite parent bodies. Proc. Natl. Acad. Sci. USA 106: 5487-5492 [Pubmed]
Hajos ZG, Parrish DR (1974) Asymmetric synthesis of bicyclic intermediates of natural product chemistry. J. Org. Chem. 39:1615-1621 [Abstract]
Hanczyc, MM, Fujikawa, SM, Szostak, JW (2003) Experimental models of primitive cellular compartments: Encapsulation, growth and division. Science 302:618-622 [Pubmed] [Full article on the Szostak lab publication page]
Hanczyc MM, Szostak JW (2004) Replicating vesicles as models of primitive cell growth and division. Curr. Opin.Chem. Biol. 8:660-664 [Pubmed] [Full article on the Szostak lab publication page]
Hanczyc MM, Mansy SS, Szostak JW (2007) Mineral Surface Directed Membrane Assembly. Orig. Life Evol. Biosph. 37:67-82 [Pubmed] [Full article on the Szostak lab publication page]
Hazen, RM, Filley TR, Goodfriend GA (2001) Selective adsorption of L- and D-amino acids on calcite: Implications for biochemical homochirality. Proc. Natl. Acad. Sci. USA 98:5487-5490 [Pubmed] [Free Full Text]
Hazen RM, Boctor N, Brandes JA, Cody GD, Hemley RJ, Sharma A, Yoder Jr HS (2002) High pressure and the origin of life. JPCM 14: 11489-11494 [Abstract]
Hazen RM (2005) Gen-e-sis: The Scientific Quest for Lifes Origins, Joseph Henry Press
Hitz TH, Luisi PL (2004) Spontaneous onset of homochirality in oligopeptide chains generated in the polymerization of N-carboxyanhydride amino acids in water. Orig. Life Evol. Biosph. 34:93-110 [Pubmed]
Huang W, Ferris JP (2006) One-step, regioselective synthesis of up to 50-mers of RNA oligomers by montmorillonite catalysis. J. Am. Chem. Soc. 128: 8914-8919 [Pubmed]
Huber C, Wächtershäuser G (1997) Activated acetic acid by carbon fixation on (Fe,Ni)S under primordial conditions. Science 276:245-247 [Pubmed]
Huber C, Eisenreich W, Hecht S, Wächtershäuser G (2003) A possible primordial peptide cycle. Science 301: 938-940 [Pubmed]
Huber C, Wächtershäuser G (2006) alpha-hydroxy and alpha-amino acids under possible Hadean, volcanic origin-of-life conditions. Science 314:630-632 [Pubmed]
Johnson AP, Cleaves HJ, Dworkin JP, Glavin DP, Laczano A, Bada JL (2008) The Miller volcanic spark discharge experiment. Science 322:404 [Pubmed]
Johnston WK, Unrau PJ, Lawrence MS, Glasner ME, Bartel DP (2001) RNA-catalyzed RNA polymerization: accurate and general RNA-templated primer extension. Science 292: 1319-1325 [Pubmed]
Joyce GF (2002) The antiquity of RNA-based evolution. Nature 418:214221 [Pubmed]
Kauffman, Stuart. (1993) Origins of Order: Self-Organization and Selection in Evolution, Oxford University Press [Amazon]
Kasting JF, Zahnle KJ, Walker JCG (1983) Photochemistry of methane in the earth's early atmosphere. Precambrian Res. 20: 121-148 [Abstract]
Klussmann M, Iwamura H, Mathew SP, Wells DH Jr, Pandya U, Armstrong A, Blackmond DG (2006) Thermodynamic control of asymmetric amplification in amino acid catalysis. Nature 441: 621-623 [Pubmed]
Klussmann M, Toshiko I, White AJP, Armstrong A, Blackmond DG (2007) Emergence of solution-phase homochirality via crystal engineering of amino acids. J. Am. Chem. Soc. 129: 7657-7660 [Pubmed]
Kojo S, Uchino H, Yoshimura M, Tanaka K (2004) Racemic D,L-asparagine causes enantiomeric excess of other coexisting racemic D,L-amino acids during recrystallization: a hypothesis accounting for the origin of L-amino acids in the biosphere. Chem. Commun. (2004): 2146-2147 [Pubmed]
Koonin EV (2007) An RNA-making reactor for the origin of life. Proc. Natl. Acad. Sci. USA 104: 9105-9106 [Free Full Text]
Koonin EV, Novozhilov AS (2009) Origin and evolution of the genetic code: the universal enigma. IUBMB Life 61:99-111 [Pubmed]
Koonin EV, Martin W (2005) On the origin of genomes and cells within inorganic compartments. Trends Genet. 21:647-654 [Pubmed]
Kuhn WR, Atreya SK (1979) Ammonia photolysis and the greenhouse effect in the primordial atmosphere of the earth. Icarus 37: 207-213 [Pubmed]
Kun A, Santos M, Szathmary E (2005) Real ribozymes suggest a relaxed error threshold. Nat. Genet. 37:1008-1011 [Pubmed]
Lee DH Severin K, Yokobayashi Y, Ghadiri MR (1997) Emergence of symbiosis in peptide self-replication through a hypercyclic network. Nature 390:591-594 [Pubmed]
Lee DH Severin K, Yokobayashi Y, Ghadiri MR (1998) Erratum: Emergence of symbiosis in peptide self-replication through a hypercyclic network Nature 394:101
Leman L, Orgel LE, Ghadiri MR (2004) Carbonyl sulfide-mediated prebiotic formation of peptides. Science 306: 283-286 [Pubmed]
McBride JM, Tully JC (2008) Physical chemistry: Did life grind to a start? Nature 452:161-162 [Pubmed]
Mansy SS, Schrum JP, Krishnamurthy M, Tobé S, Treco DA, Szostak JW (2008) Template-directed synthesis of a genetic polymer in a model protocell. Nature 454:122-125 [Pubmed] [Full article on the Szostak lab publication page]
Mansy SS, Szostak JW (2008) Thermostability of model protocell membranes. Proc. Natl. Acad. Sci. USA 105: 13351-13355 [Pubmed] [Free Full Text]
Martin W, Russell MJ (2003) On the origins of cells: a hypothesis for the evolutionary transitions from abiotic geochemistry to chemoautotrophic prokaryotes, and from prokaryotes to nucleated cells. Philos. Trans. R. Soc. Lond. B. Biol. Sci. 358:59-83 [Pubmed] [Free Full Text]
Miller SL (1953) A production of amino acids under possible primitive earth conditions. Science 117 (1953): 528-529 [PDF]
Morowitz HJ, Kostelnik JD, Yang J, Cody GD (2000) The origin of intermediary metabolism. Proc. Natl. Acad. Sci. USA 97: 7704-7708 [Pubmed] [Free Full Text]
Noorduin WL, Izumi T, Millemaggi A, Leeman M, Meekes H, Van Enckevort WJ, Kellogg RM, Kaptein B, Vlieg E, Blackmond DG (2008) Emergence of a single solid chiral state from a nearly racemic amino acid derivative. J. Am. Chem. Soc. 130: 1158-1159
Orgel LE (1998) The origin of life a review of facts and speculations. Trends Biochem. Sci. 23: 491-495 [Pubmed]
Orgel LE (2000) Self-organizing biochemical cycles. Proc. Natl. Acad. Sci. USA 97: 12503-12507 [Pubmed] [Free Full Text]
Orgel LE (2004) Prebiotic Chemistry and the Origin of the RNA World. Crit. Rev. Biochem. Mol. Biol. 39: 99-123 [Pubmed]
Orgel LE (2006) Geothermal Synthesis and Metabolism. Astrobiology 6: 297-298
Pavlov AA, Kasting JF, Brown LL, Rages KA, Freedman R (2000) Greenhouse warming by CH4 in the atmosphere of early Earth. J. Geophys. Res. 105: 11981-11990 [Pubmed]
Pizzarello S, Weber AL (2004) Prebiotic amino acids as asymmetric catalysts. Science 303:1151
Plankensteiner K, Reiner H, Rode BM (2005) Stereoselective differentiation in the salt-induced peptide formation reaction and its relevance for the origin of life. Peptides 26:535-541 [Pubmed]
Poole AM (2006) Getting from an RNA world to modern cells just got a little easier. BioEssays 28:105-108 [Pubmed]
Powner MW, Gerland B, Sutherland JD (2009) Synthesis of activated pyrimidine ribonucleotides in prebiotically plausible conditions. Nature 459: 239-242 [Pubmed]
Rajamani S, Vlassov A, Benner S, Coombs A, Olasagasti F, Deamer D (2008) Lipid-assisted synthesis of RNA-like polymers from mononucleotides. Orig. Life Evol. Biosph. 38:57-74 [Pubmed]
Rees
M (2001) Just Six Numbers: The Deep Forces that Shape the Universe,
Basic Books [Amazon]
Ricardo A, Carrigan MA, Olcott AN, Benner SA (2004)
Borate minerals stabilize ribose. Science 303:196
Robertson MP, Knudsen SM, Ellington AD (2004) In vitro selection of ribozymes dependent on peptides for activity. RNA 10:114127 [Pubmed] [Free Full Text]
Robinson R (2005) Jump-Starting a
Cellular World: Investigating the Origin of Life, from Soup to Networks. PLoS
Biol 3(11): e396 [Free
Full Text]
Russell MJ, Martin W (2004) The rocky roots of the
acetyl-CoA pathway. Trends Biochem. Sci. 29: 358-363 [PDF]
Sagan C, Chyba C (1997) The early faint sun paradox: organic shielding of
ultraviolet-labile greenhouse gases. Science 276:1217-1221[Pubmed]
Saghatelian A, Yokabayashi Y, Soltani K, Ghadiri MR (2001) A chiroselective
peptide replicator. Nature 409:797-801 [Pubmed]
Schaefer L, Fegley Jr. B (2007) Outgassing of ordinary chondritic material and some of its implications for the chemistry of asteroids, planets, and satellites. Icarus 186:462-483 [Abstract] [PDF]
Schwendinger MG, Rode BM (1989) Possible role of copper and sodium chloride in prebiotic evolution of peptides. Analytical Science 5: 411-444 [Abstract] [Free Full Text]
Smith E, Morowitz HJ (2004) Universality in intermediary metabolism. Proc. Natl. Acad. Sci. USA 101: 13168-13173 [Pubmed] [Free Full Text]
Smolin L (1999) The Life of the Cosmos, Oxford University Press [Amazon]
Susskind L (2006) The Cosmic Landscape: String Theory and the Illusion of Intelligent Design, Back Bay Books [Amazon]
Szostak JW, Bartel DP, Luisi PL (2001) Synthesizing Life. Nature 409: 387-390 [Full article on the Szostak lab publication page]
Szostak JW (2009) Systems chemistry on early Earth. Nature 459: 171-172
Tamura K, Schimmel P (2004) Chiral-selective aminoacylation of an RNA minihelix. Science 305:1253 [Pubmed]
Tamura K, Schimmel P (2006) Chiral-selective aminoacylation of an RNA minihelix: Mechanistic features and chiral suppression. Proc. Natl. Acad. Sci. USA 103:13750-13752 [Pubmed] [Free Full Text]
The RNA World. (2006) Edited
by: Gesteland RF, Czech TR, Atkins JF. Cold Spring Harbor Laboratory Press
[Pubmed]
Tian F, Toon OB, Pavlov AA, De Sterck H (2005) A hydrogen-rich early Earth
atmosphere. Science 308: 1014-1017 [Pubmed]
Viedma C (2001) Enantiomeric crystallization from DL-aspartic and DL-glutamic
acids: implications for biomolecular chirality in the origin of life. Orig.
Life Evol. Biosph. 31: 501-509 [Pubmed]
Viedma C, Ortiz JE, de Torres T, Izumi T, Blackmond DG (2008) Evolution of
solid phase homochirality for a proteinogenic amino acid. J. Am. Chem. Soc.
130:15274-15275 [Pubmed]
Wächtershäuser G (1988) Before enzymes and
templates: theory of surface metabolism. Microbiol.
Rev. 52: 452-484 [Free
Full Text]
Wächtershäuser G (1990) Evolution of the
first metabolic cycles. Proc. Natl. Acad. Sci. USA 87: 200-204 [Pubmed] [Free
Full Text]
Walker JC, Brimblecombe P (1985) Iron and sulfur in the pre-biologic ocean. Precambrian
Res. 28:205-222 [Pubmed]
Wedyan M, Preston MR (2005) Isomer-selective
adsorption of amino acids by components of natural sediments. Environ. Sci.
Technol. 39:2115-2119 [Pubmed]
Weissbuch I, Bolbach G, Leiserowitz L, Lahav M (2004) Chiral
amplification of oligopeptides via polymerization in two-dimensional
crystallites on water. Orig. Life Evol. Biosph. 34:79-92 [Pubmed]
Wolf YI, Koonin EV (2007) On the origin of the translation system and the
genetic code in the RNA world by means of natural selection, exaptation, and
subfunctionalization. Biol. Direct 2:14 [Pubmed] [Free Full Text]
Zaher HS, Unrau PJ (2007) Selection of an improved RNA polymerase ribozyme with
superior extension and fidelity. RNA 13: 1017-1026 [Pubmed] [Free Full Text]
Zahnle KJ (1986) Photochemistry of methane and the formation of hydrocyanic
acid (HCN) in the earth's early atmosphere. J. Geophys. Res. 91:
2819-2834 [Abstract]
Zhang L, Peritz A, Meggers E (2005) A
simple glycol nucleic acid. J. Am.
Chem. Soc. 127:41744175 [Pubmed]
Zhang XV, Martin ST (2006) Driving parts
of Krebs cycle in reverse through mineral photochemistry. J. Am. Chem. Soc.
128: 16032-16033 [Pubmed]
Zhu TF, Szostak JW (2009) Coupled Growth and Division of Model Protocell
Membranes. J. Am. Chem. Soc. 131: 5705-5713 [Pubmed] [Full article on the Szostak lab
publication page]
[Back to the Abiogenesis FAQs]

| Home Page | Browse | Search | Feedback | Links |
| The FAQ | Must-Read Files | Index | Creationism | Evolution | Age of the Earth | Flood Geology | Catastrophism | Debates |